Table of Contents
Estimate of Time Required: 20 Minutes
This module utilizes Electronic case report forms (eCRFs) which you will create using DFsetup without a PostScript® or PDF file. eCRFs do not contain barcodes and are therefore only suitable for entry via EDC.
-
Start DFATK as described in Module 1 (Installation & Initialization).

-
Click followed by to begin this module.
-
This module requires user
data_managerto have exclusive access to the 254 Acceptance Test Study database. The Control Panel must be closed. Click .
-
Start DFsetup, login as user
data_managerand choose 254 Acceptance Test Study as described in Section 1.8, “How to start DFsetup”. Choose Exclusive Access and click . Exclusive Access allows you to create and modify plates, styles and fields, and to define all setup configuration files.
![[Note]](../../imagedata/note.png)
Note The main DFsetup window displays the last plate to be active in the user's previous DFsetup session. For this reason, you may see a different plate from what is displayed in the above illustration.
-
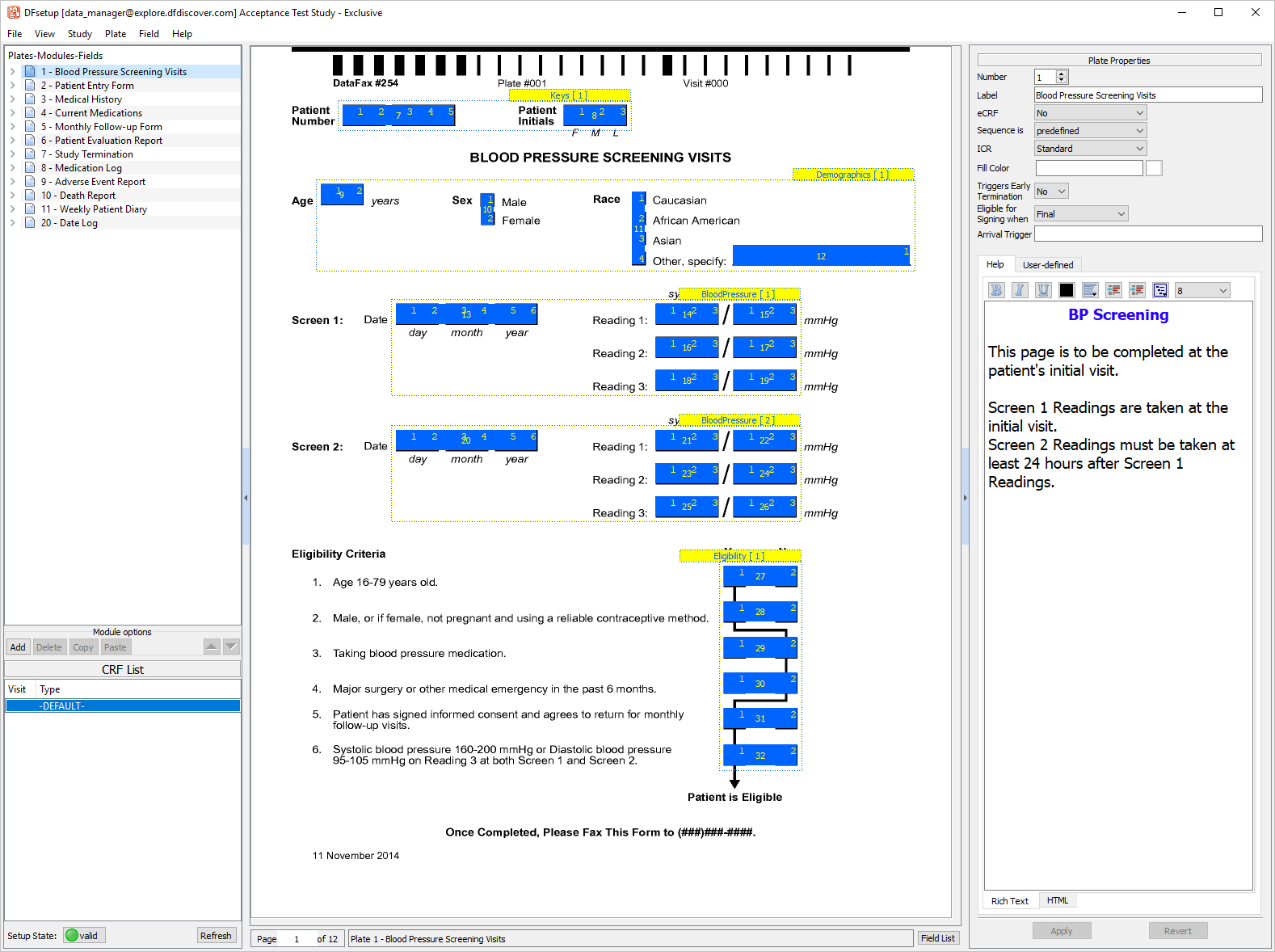
Select > . In the Add eCRF Plate dialog enter a New Plate Number of 21. Click .

In the Plate Properties set:
Label =
Lab ResultseCRF = Yes
Sequence is = predefined
Triggers Early Termination = No
Eligible for Signing When = Final
Click .

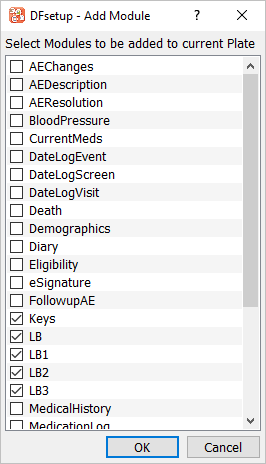
Within Plates-Modules-Fields Module options Click . Check Keys, LB, LB1, LB2, LB3 modules, click .
-
Double-click the Keys [1] module to expand the list of fields. Using your mouse, click and drag the SUBJID and PINIT fields onto the 21: Lab Results eCRF.

-
Click LB [1] Module, drag the module onto the eCRF, select LBORRES_n, LBNRIND and LBCLSIG_c fields by holding Shift and clicking on the fields. Select > .

-
Click LB1 [1] Module, drag the module onto the eCRF. Select > .

-
Click LB2 [1] Module, drag the module onto the eCRF. Select > .
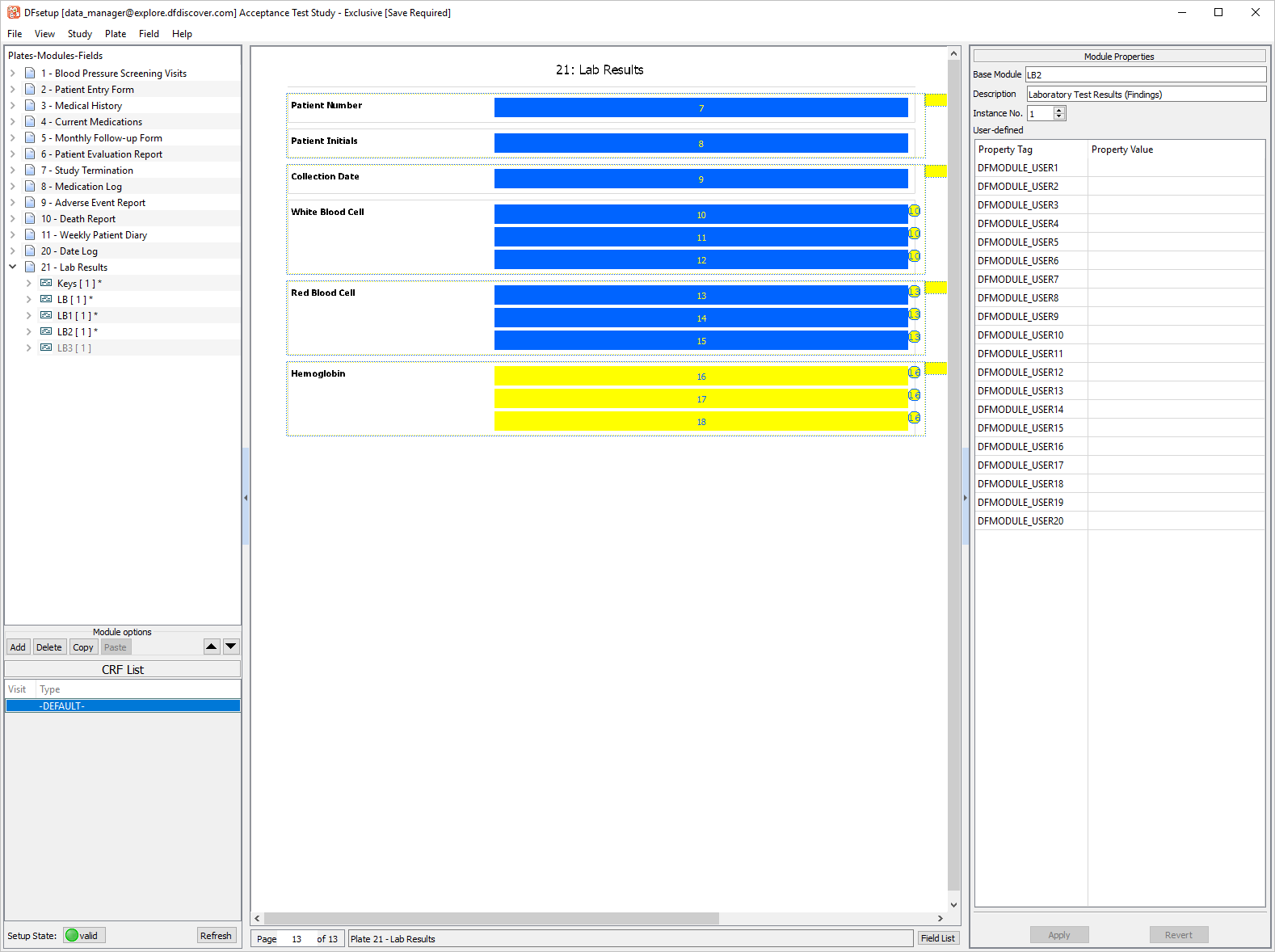
-
Click LB3 [1] Module, drag the module onto the eCRF. Select > .
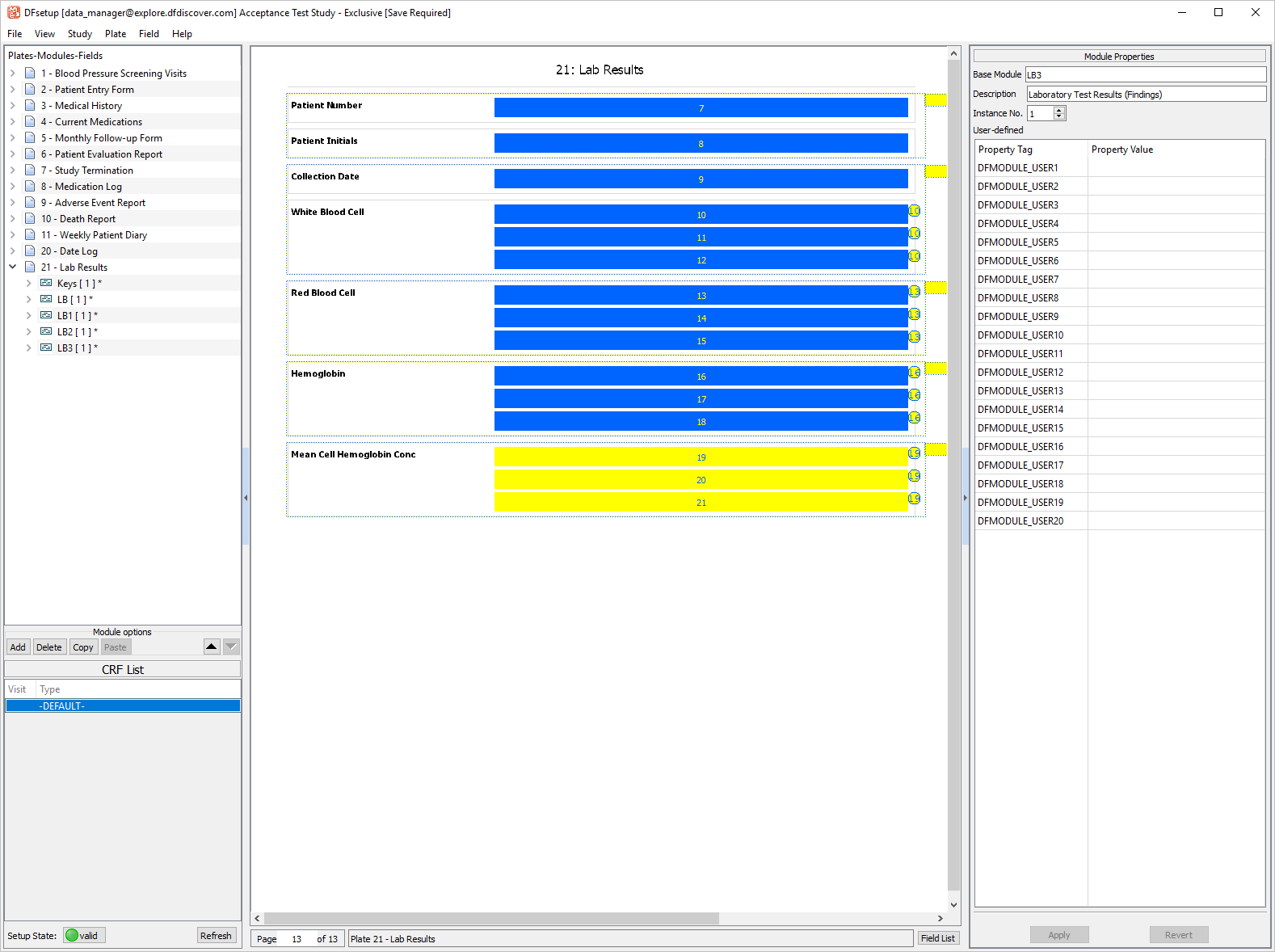
-
Select > .
-
Select > to close DFsetup.
-
Start DFexplore, login as user
site_monitorand choose 254 Acceptance Test Study as described in Section 1.7, “How to start DFexplore”. -
Click Site 99 - Hospital #99 > Subject 99008 > 1: Baseline > 21: Lab Results to make it the active page.
![[Note]](../../imagedata/note.png)
Note DFexplore stores previously defined user preferences. If
site_monitorpreviously used DFexplore and modified user preferences, you may see a different background than what is illustrated below.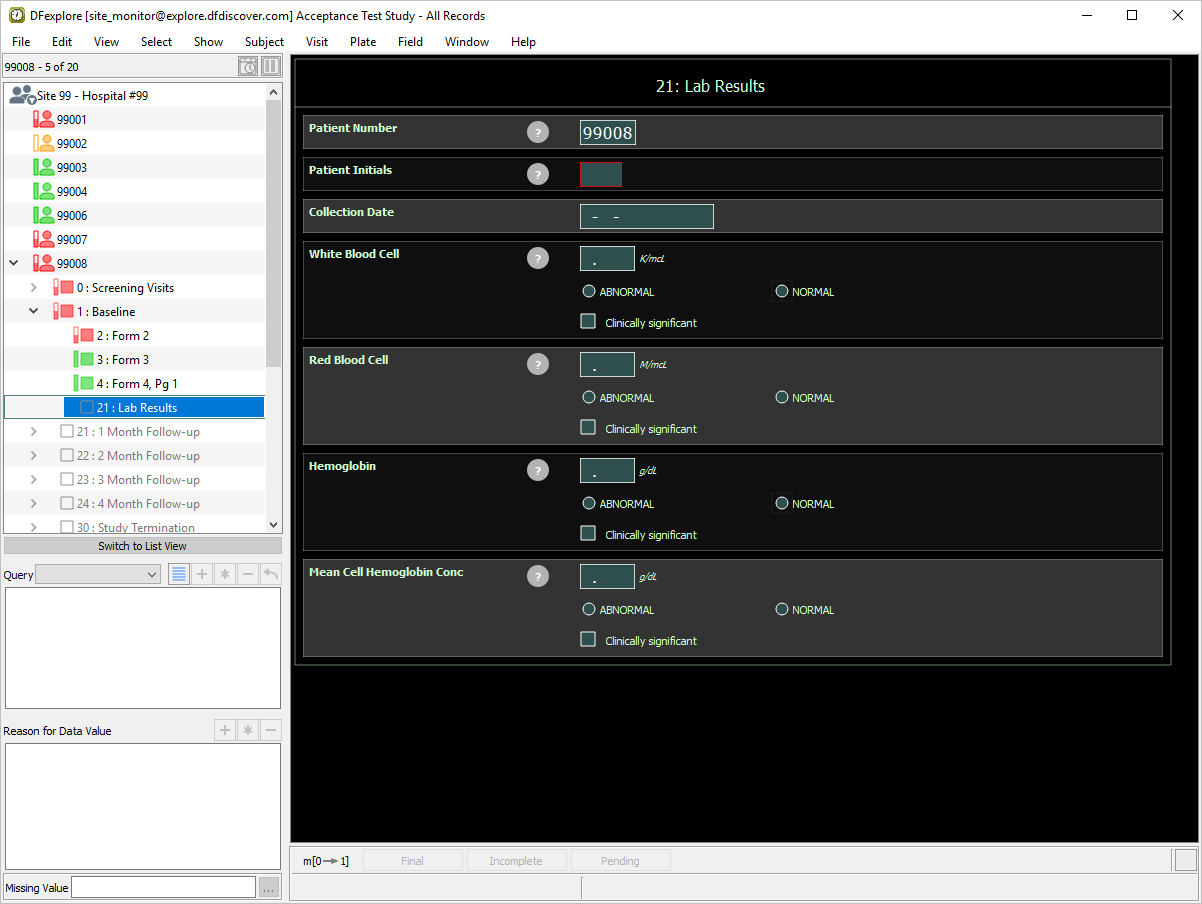
-
Select > , click Background Options to expand the preferences. Click Color then to apply the changes.

-
Select > > Data Window, within eCRF background color, click
 to open the Select Color window.
to open the Select Color window.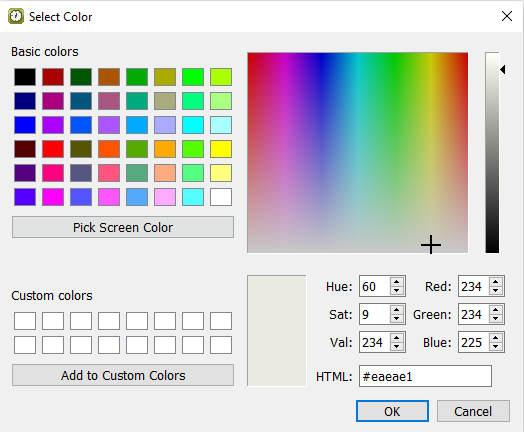
-
Enter HTML:
#66EEEE.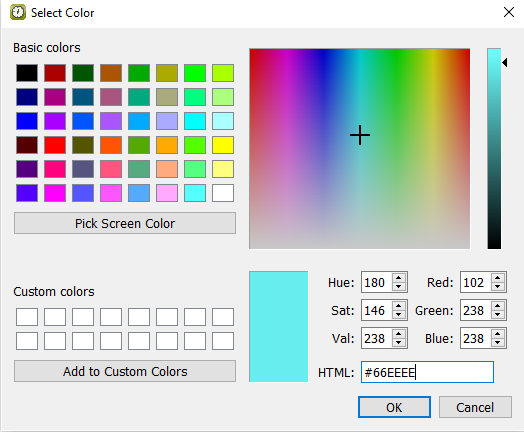
-
Click to close the Select Color window, click to apply your color preferences.
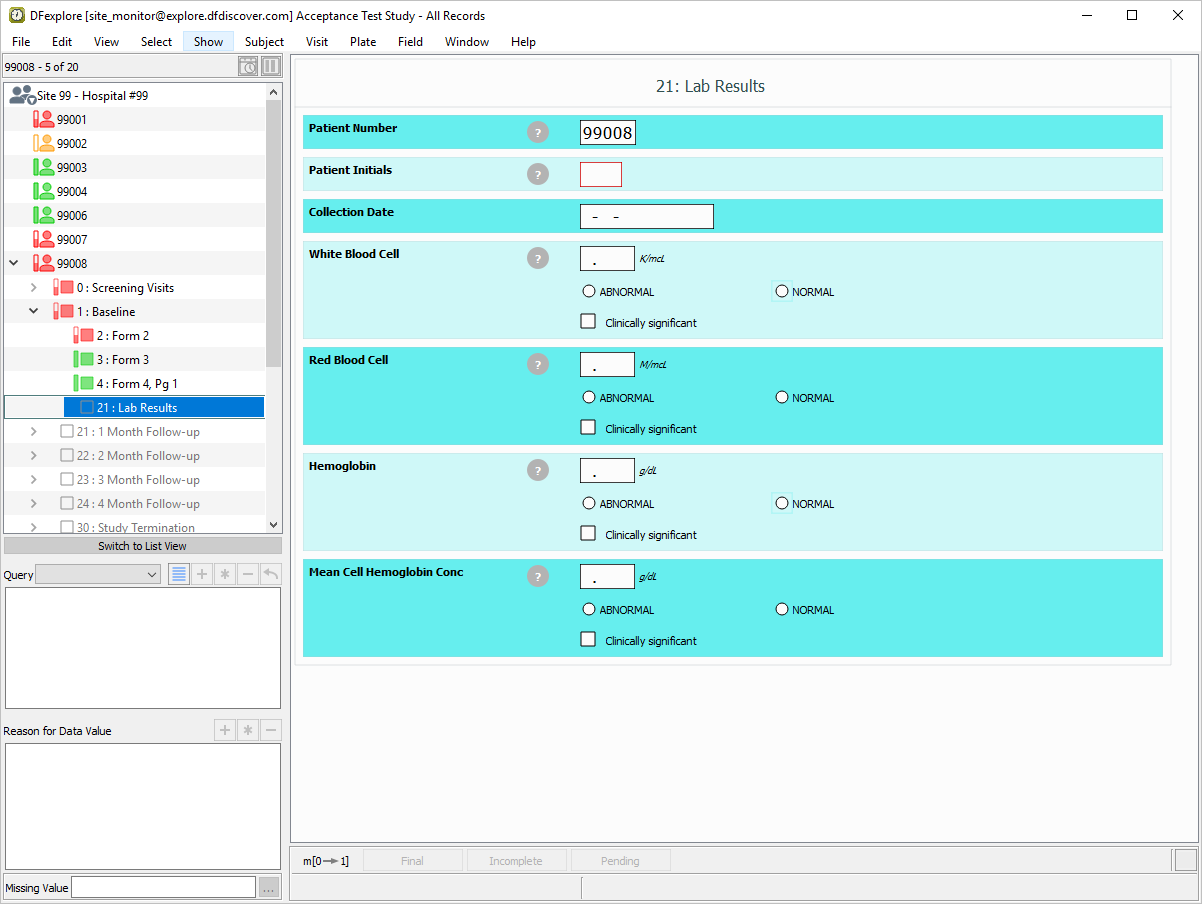
-
Click
 within the Patient Initials field to display the field level help. Click
within the Patient Initials field to display the field level help. Click  to close the help dialog.
to close the help dialog.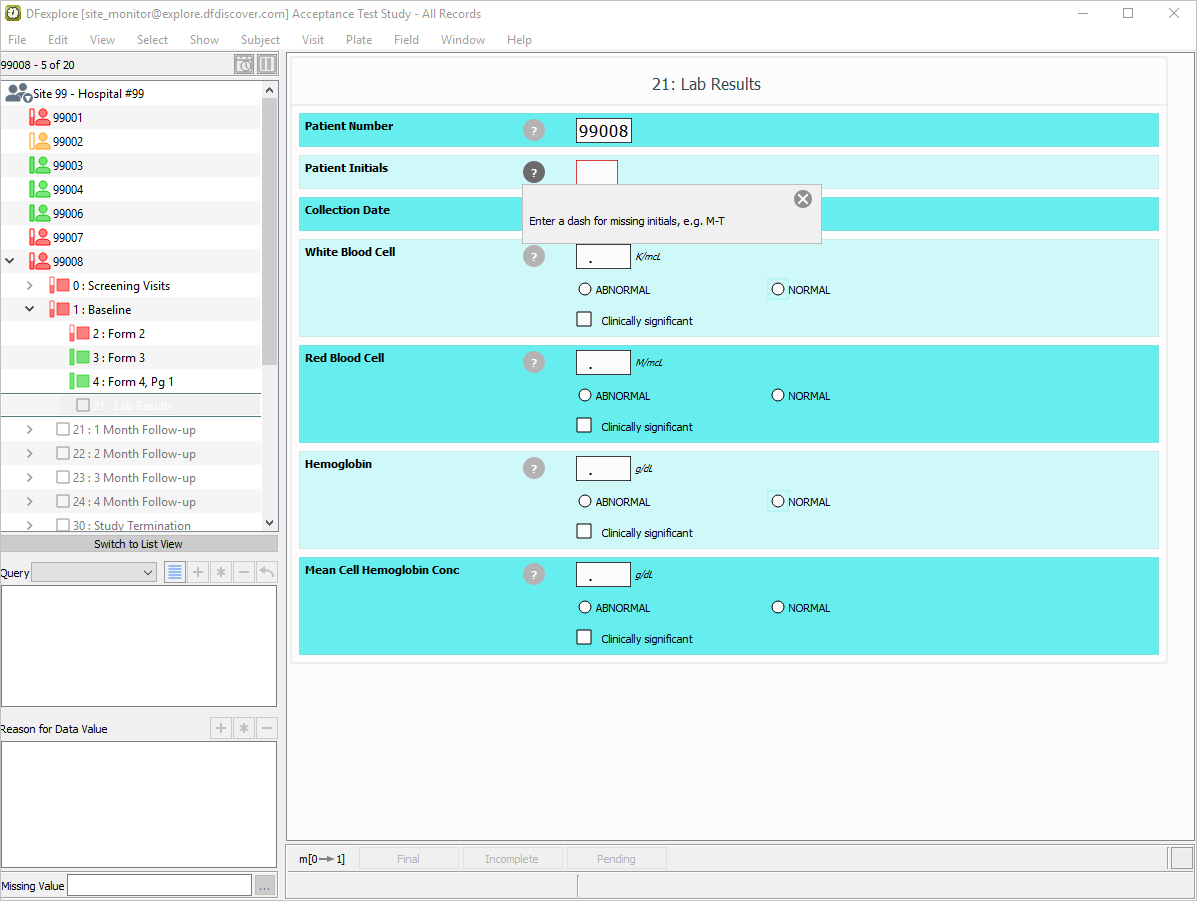
-
With subject 99008 21: Lab Results as the current eCRF, click Patient Initials. The initials are pre-filled as a result of the edit check
SetInitdescribed in Module 8. Image Data Entry. Set:Collection Date =
27-SEP-2018White Blood Cell =
18.8, Check (enable) Abnormal.Red Blood Cell =
4.7, Check (enable) NormalHemoglobin =
18.5, Check (enable) NormalMean Cell Hemoglobin Conc =
40.2, Check (enable) Normal
Click to save the data record.

-
Select > to close DFexplore.
Start DFATK as described in Module 1 (Installation & Initialization).
-
Click to confirm successful completion of Module 4. eCRF Creation.